Abstract
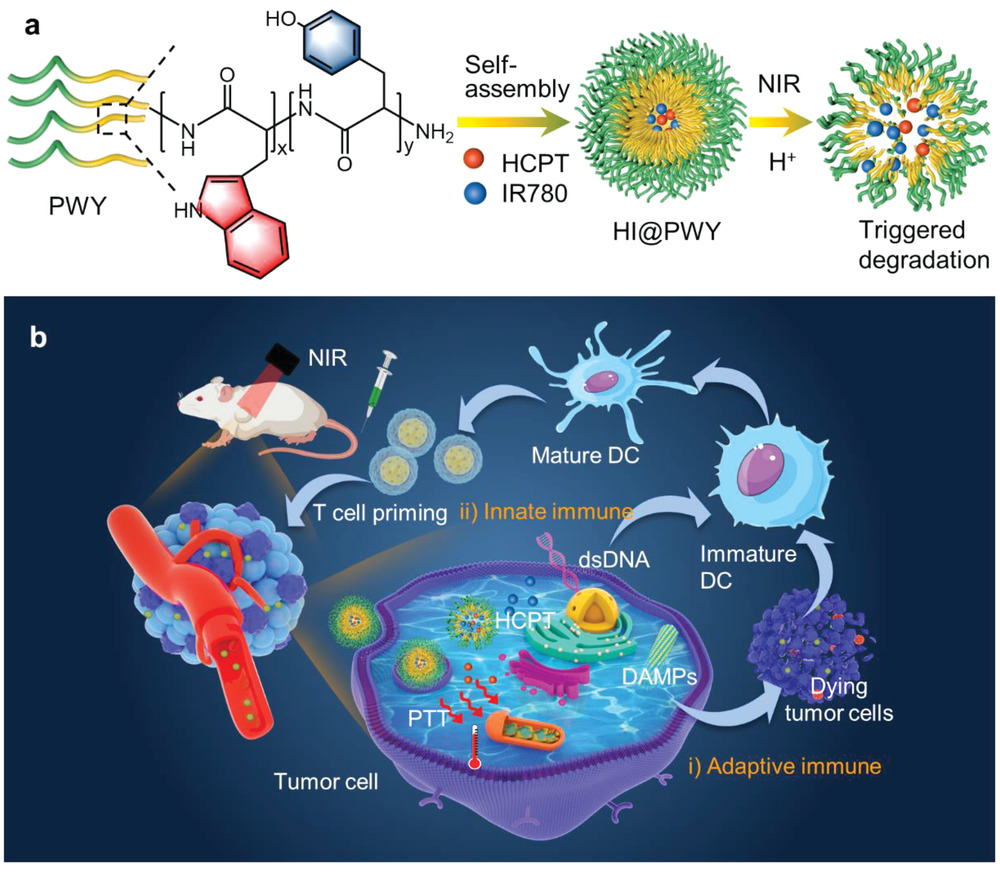
As a promising cancer treatment modality that has emerged, photothermal therapy can harness antitumor immunity by triggering immunogenic cell death (ICD) in addition to direct cell ablation. However, the immuno-stimulation induced by PTT alone is insufficient to achieve satisfactory cancer eradication, especially in immunologically “cold” tumors due to their harsh immunosuppressive microenvironment. Effective activation of the innate immune system is indispensable to boost a robust adaptive antitumor immune response typically initiated by dendritic cells (DCs). Herein the above issues are addressed by constructing an environmentally responsive supramolecular nanoself-assembly (PSAs) derived from a novel polypeptide-based block copolymer, which is capable of co-load photothermal immunomodulators efficiently under structure-guided π–π stacking interactions. In the murine model of 4T1 xenograft tumors, the fabricated PSAs with payloads trigger both adaptive and innate immune responses in situ through activation of ICD as well as STING-dependent signal pathway. The findings reveal a new mechanism of harnessing photothermal therapy toward immunologically “cold” tumors.
https://doi.org/10.1002/adhm.202202017